Tumor Grades
Grading helps us understand how aggressive a tumor is and if the tumor has spread, and if so, how far. There are four grades of brain tumors, and the higher the grade the more malignant the tumor. Generally speaking, the lower the tumor grade, the better the chance of recovery. Tumor grading helps the doctor, patient, and caregivers/family members to better understand the patient’s condition. It also helps the doctor plan treatment and predict outcome.
Below are description of the various tumor grades, based on the World Health Organization (WHO) grading system:
- Grade I: These are the least malignant tumors and are usually associated with long-term survival. They grow slowly and have an almost normal appearance when viewed through a microscope. Surgery alone may be an effective treatment for this grade tumor.
- Grade II: These tumors are slow-growing and look slightly abnormal under a microscope. Some can spread into nearby normal tissue and recur, sometimes as a higher grade tumor.
- Grade III: These tumors are, by definition, malignant although there is not always a big difference between grade II and grade III tumors. The cells of a grade III tumor are actively reproducing abnormal cells, which grow into nearby normal brain tissue. These tumors tend to recur, often as a grade IV.
- Grade IV: These are the most malignant tumors. They reproduce rapidly, can have a bizarre appearance when viewed under the microscope, and easily grow into nearby normal brain tissue. These tumors form new blood vessels so they can maintain their rapid growth. They also have areas of dead cells in their centers. The glioblastoma multiforme is the most common example of a grade IV tumor.
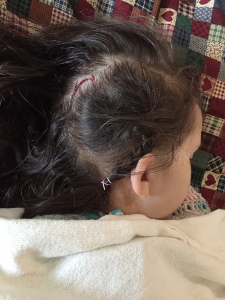
Ok, I’m just going to say it: The WHO’s description is misleading and has created a ho-hum attitude about Grade I brain tumors that is frustrating and offensive. Kiara’s tumor is a Grade I pilocytic astrocytoma and it is not a friendly little tumor that you scoop out and it goes away. It has wreaked havoc on her body — permanently compromised her balance and taken her vision. Plus, it is against (grabbing into) her brain stem, so this is inoperable and life-threatening. She has endured 68 weeks of chemotherapy treatments and will have to face the consequences of what that may have done to her body. The paradigm about Grade I battles needs to be changed.
Pathology
Pathology is a branch of medical science primarily concerning the cause, origin, and nature of disease. It involves the examination of tissues, organs, bodily fluids, and autopsies in order to study and diagnose disease. In Kiara’s case, they sent her brain tumor to the pathology lab, where they run tests to determine the type of tumor and the grade. Biochemists or chemical pathologists examine different substances found in the tumor that can help understand its origin and behavior, so that doctors know how to treat it. Pathologists may also study the molecular and genetic profiles of a tumor to determine different tumor subtypes. Neuro-oncologists may use the genetic studies to inhibit the growth or recurrence of a tumor. In Kiara’s case, I’m interested in learning more about BRAF status in pilocytic astrocytomas, which understanding is way above my pay grade right now, but keeping it in my back pocket for future discussions with her oncologist.
Ventriculoperitoneal Shunt
A ventriculoperitoneal (VP) shunt is a medical device that is implanted by a neurosurgeon to treat hydrocephalus and drain excess cerebrospinal fluid (CSF) when the brain ventricles have accumulated too much, potentially causing pressure on the brain. It sits below the skin behind the ear, with one tube gathering CSF from the brain and another tube draining the CSF down into the abdomen, where it is reabsorbed by the body.
Kiara’s vision continued to worsen, so the hypothesis was that there may be increased ICP on her optic nerves. Even though she already had a successful ETV, her neurosurgeon implanted a v/p shunt so that we could be assured that her brain was not a “crock pot of pressure” that could cause her further problems down the line. Although we had been professional shunt-dodgers for a year, we finally consented to the device. Her shunt was manufactured by Certas and it is programmable.
Port
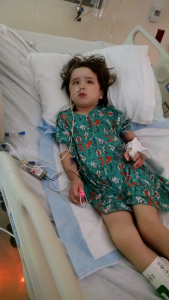
A surgeon implants a port in the chest just under the skin. It is a small disc (kind of like a little pin cushion) made of plastic or metal about the size of a quarter that has a soft thin tube called a catheter connecting the port to a large vein near the heart. Chemotherapy and other IV medications are given through a special needle that fits into the port. Blood can also be drawn through the port to test ANCs and other panels. Kiara’s port is accessed every Thursday during her oncology appointment. After her chemo roadmap is finished, she will go to the clinic 1-2x per month to have her port flushed. After a couple MRIs over the course of 3 or so months and if her scans are stable then her port can be surgically removed.
Posterior Fossa Syndrome
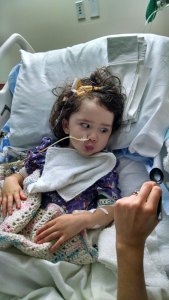
I was not prepared for how Kiara would look after her first major tumor resection. Even though her neurosurgeon discussed the likelihood of her having posterior fossa syndrome, it’s difficult to even type the experience. She was trapped in what can only be described as “neuro storms.” Her mouth and tongue would twitch and her eyes jittered separately and in different directions. She was completely unresponsive to me or any of her caregivers like she was in a coma or far, far away. I knew she was in there and probably scared and hurting, and I didn’t leave her side. I thought she may die at any second because her brain and body were just not right. Her head was strongly tilted to the right and her right eye was completely inward. Her lips were puckered. She had tubes everywhere, including an EVD, two IVs, a feeding tube, and a catheter, plus the pulse monitor and blood pressure cuff. Monitors constantly beeped and alarms went off frequently. It was heartbreaking and devastating.
Posterior Fossa Syndrome is a post-operative condition that affects patients that have had surgery on the back side of the base of the skull that contains the brain stem and cerebellum. Symptoms usually appear 1–5 days after surgery. Most symptoms improve in weeks or months, but some symptoms may continue for years following surgery. The symptoms of posterior fossa syndrome include problems with speech and language, motor skills, and mood changes; however, symptoms can be different for each person.
Over the course of a couple of weeks, Kiara slowly regained control of her face, eyes, and body. Speech therapists helped her relearn how to swallow, eat, and speak; physical therapists helped her regain gross motor skills. There was nothing more terrifying than watching her “neuro storms” after her first major resection, and I was so very grateful that she did not have posterior fossa syndrome after her second craniotomy.